
Galvanic reaction between dissimilar metals is an enormous topic—and for good reason. Galvanic reactions, also known as bimetallic corrosion, can wreak havoc on projects that are expensive and meant to last for a long time. One well known example is the Statue of Liberty. In 1980, workers discovered that structural issues were beginning to show. The statue’s internal iron support system was deteriorating with rust because the insulating layer of shellac between the iron and copper had begun to fail, which allowed for galvanic corrosion between the two dissimilar metals.
Most architects, engineers, designers and fabricators have heard of galvanic corrosion. Most are aware that it can become a serious problem, but most are not sure how it works.
It will be helpful, to first explain the science and terms commonly used on this topic.
Galvanic corrosion, named after Luigi Galvani who discovered this condition, occurs when using dissimilar metals together. To be exact, when using base metals that make contact with noble metals. Galvani discovered that chemical reactions can create electrical currents. Aptly named galvanism, this current is completed by electrolytes in the reaction itself.
What are electrolytes? Substances—in this case, metal—that share ions when exposed to water. Most water-based liquid, or even moisture, contains electrolytes. Electrolytes are divided into three types: acids, bases and salts. They act like wire conductors connecting and creating an electrical circuit between the two metals.
When the two metals electrically connect, they become polarized—think of the term, opposites attract. The positive side acts as a cathode (+) receiving ions and the negative side acts as an anode (-), losing ions and corroding.
Here is an attempt to explain this in layman terms. When one metal comes in contact with another, there is current flow between them. The current flow changes when dissimilar metals come in contact with each other; the corrosion rate of the active, or least corrosion-resistant metal, accelerates, and the inactive, or more corrosion-resistant metal, decreases. (That explained everything…right?)
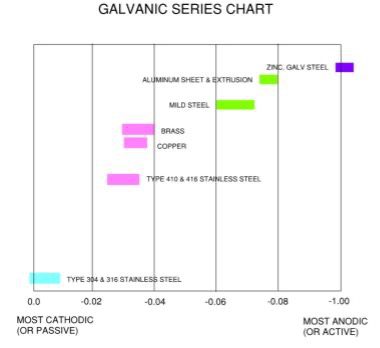
In addition, the further apart the metals are on the corrosive scale, the stronger the galvanic reaction between the two. A side note that might be obvious but worth mentioning—similar metals have little galvanic effect on each other.
There is a huge range of metals to consider, but we are going to focus on the common ones in the fabrication industry.
Metals most commonly used in fabrication include steel, stainless steel, aluminum, copper, brass and zinc.
Here is a helpful chart that lists several metals with highlighted boxes on the six listed above.
Things to remember when trying to use dissimilar metals:
One of the big gotchas is fasteners. It will be a disaster to use, for example, zinc-coated fasteners on stainless materials.
The relative surface of dissimilar metals used in a metal fabrication project should also be taken into consideration. For example, using inactive metals as the main body of the project, joined by active connectors, could cause the joints or fasteners to corrode at a quicker rate. Best-case scenario would be to connect two sections of similar metals with fasteners also made of the same metal. If using similar metals isn’t possible, a compatible metal is the next best choice.
The obvious point is that when using dissimilar metals in a fabrication project, precautions must be taken to prevent galvanic reaction.
Brass and copper are a bit in no man’s land—for example, what type of fasteners would best be used? Common practice is to use stainless steel fasteners for a couple of reasons. One, they are more passive so we are heading downward on the chart; two, if, for example, you are fastening copper panels with stainless steel fasteners, they allow for the bigger, physical square foot areas of the panels to be on the more active side of the reaction, which is also helpful.
When dissimilar metals are a concern, another great way to stop the galvanic reaction is to use inert barriers between the metals. Barriers such as paint, coating, special caulk, not-metallic washers, insulation, ice and water shield are all good examples of inert barriers that prevent the two metals from touching.
Whenever possible, the best solution when two types of metal are used together, is to use the same material. That seems like a no-brainer but often times, particularly in architectural work, pretty metals like copper and zinc, are often too soft and not rigid enough to create the necessary structural integrity.
Substituting aluminum for structural steel, for example, is not a very good solution for constructing support or strength (and also very expensive). Aluminum fasteners are another example of something that has low shear strength and, therefore, not very structurally sound.
There are ways around dissimilar metals, but it is worth the time to take a deep dive into what solutions are available and what solutions are going to truly work. As mentioned earlier, there is a large amount of misunderstanding and misinformation about this important topic.
All Metals Fabrication is an expert in the metal fabrication industry, which definitely includes understanding galvanic reactions. Our craftsmen and engineers are certified in their respective fields and dedicated trade professionals.
Learn more about us at www.allmetalsfab.com or contact us at info@allmetalsfab.com.